Evaluating Parylene Dimer Purity
When evaluating parylene dimer purity, it is first important to understand how each supplier controls and reports quality. A great way to gain an understanding of a supplier’s quality practices is to review the Certificate of Analysis that should come with every order of parylene dimer you receive. The Certificate of Analysis may include information such as:
- Identity and Appearance
- Purity
In this article we will discuss the fundamentals of each item that may be reported on your Certificate of Analysis and why it is relevant to parylene dimer. After reading this article, you should not only have a better understanding of how the tests are performed, but also how to analyze the resulting data. Once you’re able to accurately interpret the Certificate of Analysis, you will be able to gain a much better insight into the quality control that your parylene dimer supplier provides.
The criteria stated on your Certificate of Analysis is influenced by a variety of tests, including melting point and purity testing. Before diving into the specifics of how each of those tests is performed, we will first discuss dimer types C, N, and F and how each can be visually identified.
Parylene Dimer Chemical Identity and Appearance
In its physical form parylene dimer is a white to off-white, crystalline powder. In general, the more yellow a parylene dimer is, the less pure it will be. Furthermore, when a parylene dimer is a lower purity, it may clump together and be less like a free-flowing powder, an indication that residual solvents may have been left behind from the chemical synthesis of the dimer. In comparison with a few everyday products, parylene dimers have been compared in appearance to baking soda, laundry detergent, or granulated white sugar as can be seen below.
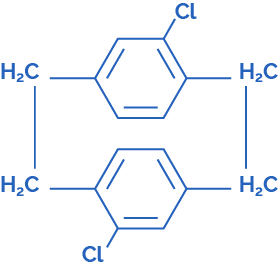
Parylene C Dimer – Dichloro[2.2]paracyclophane

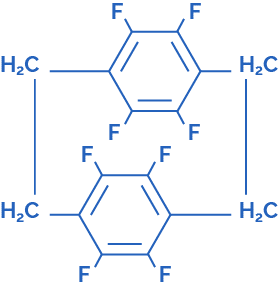
Parylene F dimer – Octofluoro[2.2]paracyclophane

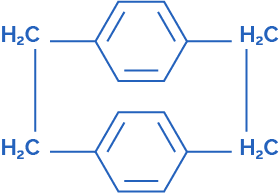
Parylene N dimer – Di-p-xylene

Parylene Dimer Purity Testing
Although being able to differentiate what parylene dimer looks like on paper and by the naked eye, it is important to understand the numerous laboratory tests that can be performed to further determine the purity of your parylene dimer. The three different laboratory tests that we’re going to discuss are:
- Melting Point
- Gas Chromatography – Mass Spectroscopy (GC-MS)
- High Performance Light Chromatography (HPLC)
Melting Point
Knowing the melting point of your parylene dimer can be useful in gaining an initial sense of your parylene dimers identity and even purity. Each variety of parylene dimer has its own specific melting point, or melting point range, which can help you identify the parylene dimer type. In addition, the melting point range can give you insight into your parylene dimers purity,
It is important to note that the value reported for melting point is often times the median temperature value of a melting point range, which begins at the temperature when the solid just begins to melt and ends when the solid has completely melted. Melting point is considered a colligative property, meaning that the results will be dependent on the concentration of different molecules within your solution, rather than the nature of the molecules.
When you have an impure dimer sample, it takes less energy, or heat, to start the phase change from a solid to a liquid. This is due to weaker intermolecular bonds between non-identical molecules within your sample, which is most likely different types of parylene dimer. Therefore, the melting point range for an impure sample will not only start at a lower temperature, but will also result in a broader range as each type of molecule goes through its phase change. .
When compared to a pure, dimer, a lower purity dimer will
have a lower median melting point and a broader melting point range.
The melting point is a simple test that can easily be performed in-house by using a melting point apparatus. While melting point apparatuses may vary in design, most will require a capillary tube to be loaded with a sample and placed into the heating source of the apparatus. This heating source will either be a heating block or an oil bath, depending on the apparatus. One of the most important variables to take into consideration when performing the melting point test, is the temperature ramp rate of the instrument being used.
The temperature ramp rate of the melting point apparatus will determine how accurate your reported results can be. If the ramp rate is too high, the tester won’t be able to accurately determine the start and stop temperatures. Although the video, below, was sped up for the sake of this article, a ramp rate of no faster than 1°C per 30 seconds is recommended when performing the melting point test on parylene dimer samples. This allows for any variation within the thermodynamic system to be detectable.
The video, below, is an example of what you would likely see when performing a melting point test. While watching the video, take note of two moments in particular:
- when melting begins
- when melting is complete.
Knowing the melting point range of your parylene dimer can help you differentiate between the various types of parylene dimers, which are visually very similar; and therefore, run the risk of accidentally being mixed. Due to the melting points of the various parylene dimer types being significantly different, performing a melting point test in-house can give you quick peace of mind before sending your dimer out to a laboratory for further testing to determine purity.
| Parylene C
Dimer Melting Point 160° – 180°C |
Parylene F
Dimer Melting Point 240 – 260 °C |
Parylene N
Dimer Melting Point 280 – 300 °C |
Once you have peace of mind that the dimer you have received is, indeed, the correct type, it’s time to send your dimer out for purity testing. There are a couple laboratory techniques, known as Chromatography, to choose from.
Chromatography
Chromatography is a laboratory technique that can be used to analyze multiple chemical compounds in a variety of situations such as, drug detection, environmental analysis, explosives investigation, and identification of unknown samples. There are a variety of chromatography techniques; however, they all function under similar basic principles of compound separation, identification, and quantification.
Gas chromatography – Mass Spectrometry, or GC-MS, and High Performance Light Chromatography, or HPLC, are two types of chromatography techniques that we will be discussing in this article, as your supplier(s) most likely use one of these two tests to determine the chemical make-up and purity of their parylene dimers. Understanding the differences between the two tests and which test your supplier chooses to use, will give you the transparency you need to be confident in your suppliers’ quality control.
Gas Chromatography – Mass Spectrometry
First, we are going to look at Gas chromatography – Mass Spectrometry, or GC-MS. GC-MS is an analytical method that combines the features of both gas-liquid chromatography and mass spectrometry to identify different substances within a volatile test sample. GC-MS requires the interaction of a mobile and stationary phase, as well as boiling points, to separate molecules, To better understand the process of GC-MS, please see the diagram below:
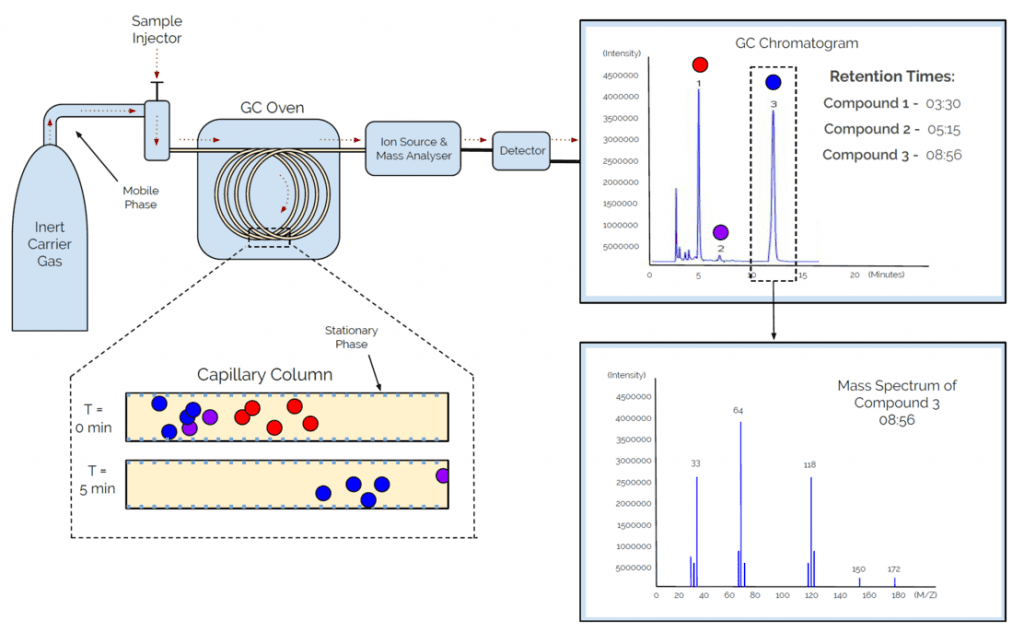
As seen in the diagram, above, the sample is injected into an inert carrier gas, which is considered the mobile phase. This mobile phase carries the sample as it enters the GC oven via a capillary column. The capillary column contains a stationary phase, which in this case is a chemical on the surface of the tube. While in the capillary column, the interaction between mobile and stationary phases occurs in conjunction with the effect of boiling point due to raised temperatures of the GC Oven.
As the mobile phase passes across the stationary phase, some of the molecules within the mobile phase begin to temporarily stick to the surface of the stationary phase. Each different type of molecule within your sample will result in a different reaction between the mobile and stationary phase, which ultimately affects what time the molecules reach the detector; also known as, the retention time. Furthermore, since we know GC-MS is a volatile technique, the boiling points of the compounds will also greatly affect retention time, as a compound with a lower boiling point will move quicker through the capillary column; therefore, making it to the detector before other compounds and lowering the retention time.
While entering the detector, the molecules are blasted with electrons, causing each molecule to break into unique fragments that turn into positively charged particles, or cations. These cations then travel through an electromagnetic field that filters the cations by mass while entering the detector to be analyzed based on their mass-to-charge ratios. A computer algorithm not only produces a chromatogram, but it also provides a mass spectrum, or MS, for each separated molecule.
The mass spectrum produced can then be used to compare and match against a library database that contains known chemical mass spectrums; therefore, providing a breakdown of all chemicals present. The results of a GC-MS test can be depicted as both a chromatogram, with retention time on the X-axis, or as a spectrum, with mass to charge ratio on the X-axis. By combining the two, we are able to identify the chemical as well as determine its concentration within your sample. Therefore, the most important information gained from your GC-MS report, is the percentage of each compound present in your sample, as this will give you purity, which is then reported on the Certificate of Analysis.
High-Performance Light Chromatography
High-Performance Light Chromatography (HPLC), which is often interpreted as “High Pressure” Light Chromatography, due to its functionality, is another type of chromatography that can be used to determine the purity of both volatile and non-volatile samples. Although both GC-MS and HPLC use the interaction between mobile and stationary phases to separate compounds, HPLC uses polarity as a separation function, while GC-MS uses volatility. To better understand the HPLC process, please see the following diagram:
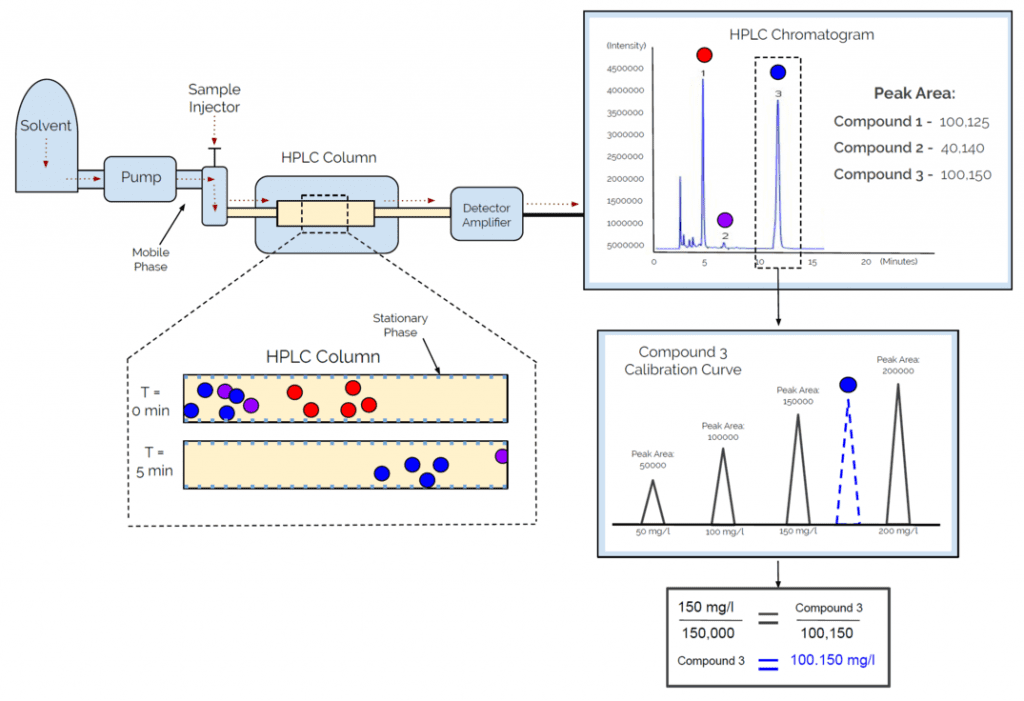
As seen in the diagram above, the mobile phase, often times made up of water and an organic solvent, is injected with a sample of varying polarities and pumped through an HPLC column. As the mobile phase is pumped through the column, the percentage of water in the mobile phase decreases over time, while the percentage of apolar solvent increases; resulting in the mobile phase becoming more apolar over time. Therefore, the compounds within the sample are able to stick to either the column or the mobile phase, depending on polarity. Eventually, all compounds will stick to the mobile phase, when their polarities match, and ultimately be eluted from the column at a particular time, called the retention time.
Once all compounds pass through the HPLC column and into the detector, which includes an ultraviolet (UV) light detector, a chromatogram is produced with the resulting intensities, or absorbance peaks, of each compound. By having a general idea of what your sample is made up of, you’re able to compare retention times and absorbance peak areas to standards in order to identify each compound present.
After identifying each compound, a calibration curves of each compound can be used to determine the actual concentration of each compound present within your sample, By combining retention times with absorbance peak areas and calibration curves, we are able to identify the chemical as well as determine its concentration within your sample. Therefore, the most important information gained from your HPLC report, is the percentage of each compound present in your sample, as this will give you purity, which is then reported on the Certificate of Analysis.
GC-MS vs HPLC, which is preferred?
When it comes to purity testing, both GC-MS and HPLC can be used to determine your parylene dimers’ purity; however, GC-MS does have its advantages when seeking the most accurate and cost-effective results. GC-MS not only works great with volatile samples, which parylene dimer is, it also results in more precise chemical composition information due to results being compared to a cataloged database.
On the other hand, HPLC uses a UV detector, which is much more relative due to the potential of one compound absorbing more strongly at a lower concentration, as compared to another compound. As discussed, HPLC also requires the results to be compared to calibrated standards that are created from pure samples. There is such a vast variety of parylene dimer types, that in order to accurately detect each variance within your sample, the UV detector response, would have to be calibrated with pure compounds of each parylene dimer type you could possibly expect.
Though both test methods can yield similar results, VSi chooses to use GC-MS when performing parylene dimer purity testing. By choosing GC-MS, VSi ensures that any trace amounts of any varying parylene dimer types can be detected and reported back to us. VSi understands that the purer your parylene dimer, the most efficient your process can be; therefore even minute variances in purity are important to us!
Choosing the Right Parylene Dimer Supplier
Choosing a supplier for your raw material can be daunting, especially when you aren’t an expert on the subject. Having the know-how to ensure that the parylene dimer you’re receiving is up to the very best quality standards can bring you peace of mind. Proper identification and acceptable purity results not only helps you confirm that your supplier has adequate quality control, but also aids in creating a cost-effective and efficient process.
A quality parylene dimer is one that has a high purity; the higher your dimer purity, the more efficient your parylene coating process can be, given that the process controls are properly set.
Highest Transparency in Purity
VSI stands by the quality and performance of its dimer. That is why we offer the industry’s only 100% satisfaction guarantee. Learn more about VSi’s family of reliable, high-performance dimers, parylene coating equipment and parylene coating and development services.