Pale skin, potato salad, parylene. What do these things have in common? Each will come to harm if left in the heat too long.
Everything in existence has an upper-temperature limit, so the selection of the type of parylene that you want to use to protect your product depends strongly upon what the temperature conditions will be. Keep reading to learn more about parylene thermal properties and their implications.
Parylene Operating Temperatures
All materials degrade, especially as the temperature increases. The same is true for parylene coatings and each has specific temperature limits that once reached, vastly shorten the usefulness of the coating. Near the end of the life of the parylene coating, it loses flexibility and becomes more brittle, can yellow, and forms cracks which lead to coating failure. This is known as a glass transition, when the parylene transitions from being flexible and salient, to brittle and opaque. Parylene glass transition temperatures depend on the type of parylene.
To mitigate the risk of any coating failures in your products due to temperature, you need to know and share with your coating provider the expectations and/or specifications of your product’s end-use environment. Parylene temperature stability values for different parylenes are collected in Table 1 below and are based on industry literature.
Table 1. Temperature stability and melting points of the parylenes based on industry literature
| Parylene-Types | Long-Term Temperature Limit (°C) Duration = ~10+ Years | Short-Term Temperature Limit (°C) Duration = ~1 Month | Melting Point Temperature (Tm) |
| Parylene N | 60 | 95 | 420 |
| Parylene C | 80 | 115 | 290 |
| Parylene D | 100 | 135 | 380 |
| Parylene F | 200 | 250 | – |
| Parylene AF-4 | 350 | 450 | >500 |
Parylene Melting Temperatures
You have bigger problems to worry about than the parylenes melting if temperatures are getting that high. The melting points for all of the parylenes are very high and are typically above that of solder and other materials on circuit boards, as seen in Table 1 above. The parylenes have glass transition temperatures where the parylene polymers transition from a more ordered/crystalline state to a more plastic/amorphous state. During this transition, when the parylene is heating or cooling, it can cause mechanical stress on interior components but the stress is very minimal and rarely affects the product.
Cracking
Protective coatings, including parylenes, work best when they’re free of pinholes, bubbles, and most importantly cracks. Exposure to heat and oxygen causes microscopic breaks in most parylene films until its severe enough to cause cracks visible to the naked eye. On a side note, the same is true for most parylene film’s exposure to ultraviolet (UV) light from the sun and oxygen.
In both cases of high temperature and oxygen or UV light and oxygen, most of the parylenes are oxidized. Carbon to oxygen bonds are added to the methylene (CH2) units in the polymer backbone: first as an alcohol (-OH), then a carbonyl (C=O), and then a carboxylic acid (-COOH) which causes a break in the polymer chain. When enough of the polymer chains have broken in such a manner, then a visible crack will form.
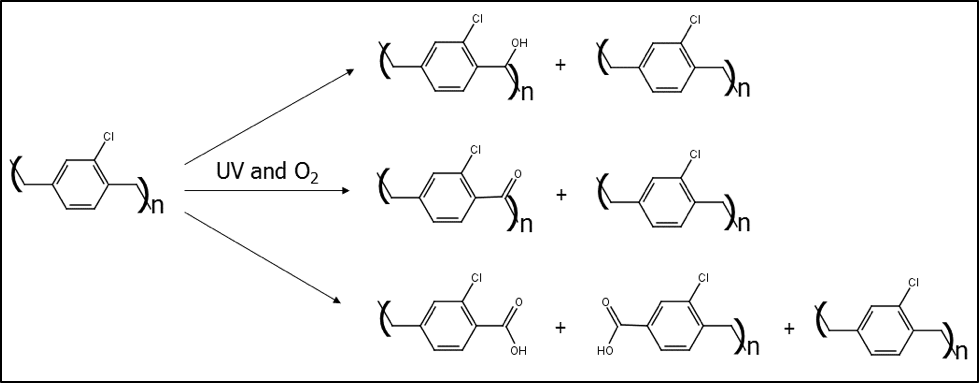
Figure 1. Schematic of the oxidation products of parylene C.
Once a crack has formed, the product will be exposed to possible contamination and corrosion. Since this oxidation and degradation mechanism involves CH2 units, the parylenes N, C, D, and F are susceptible. Only parylene AF-4 has CF2 units bridging between the rings in the polymer chain and that’s why it can survive much higher temperatures and much longer UV exposure than the other parylenes. The chemical structures of the parylenes are shown in Figure 2 below.
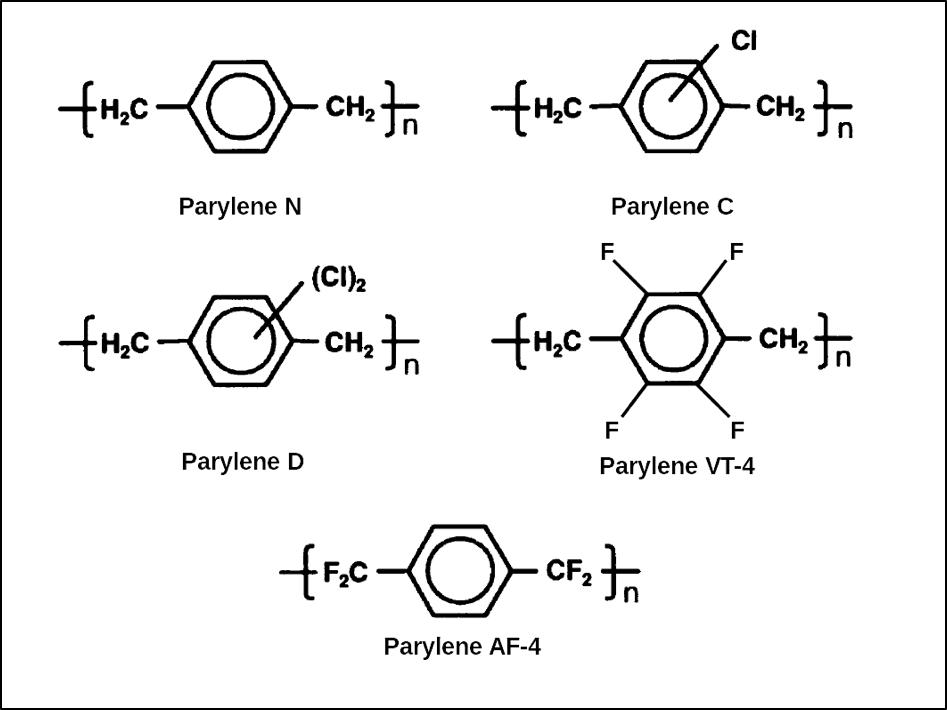
Figure 2. Chemical structures of the parylene repeat units.
Changing Color
Physical changes can sometimes represent chemical changes, as is the case with parylene as it oxidizes. Parylene will yellow as the film is exposed to heat or UV light and oxygen, a visible indicator that the coating is being changed. The clear coating takes on a slight yellow hue as it is being oxidized and will become more yellowed, similar to a very pale highlighter, but not as intense. As the film yellows, it will also develop microcracks that will grow larger until visible to the naked eye. Around the edges of the cracks the coating curls upwards and appears like dried mud or a lake bed, as illustrated in Figure 3 below. If the cracked coating is then flexed or the product is in motion, then the cracks will quickly become worse.

Figure 3. Image of cracked mud.
Expansion
As with most things, the coating and the coated product expand with higher temperatures and shrink with lower temperatures. Since parylene is typically applied as a very thin coating, there is very little stress applied to solder joints and other structures on electronics assemblies and other products.
If the rate of expansion, also known as the coefficient of thermal expansion (CTE), of the coating and the coated product aren’t close to one another, then it’s likely that cracks can form in the coating due to this mismatch.
Parylene Coating Outgassing
The epoxy-glass fiber composites of the more commonly used FR-4 style circuit boards can absorb water from the air and multiple processing steps, as well as the many other chemicals it has been exposed to. This is one reason why it’s important that any rinsing baths are properly maintained to remove most chemicals from dip processing steps.
Any adhesive, masking, staking, and related materials should be fully cured prior to coating with parylene. Also, including a mild bake out or bake out under vacuum step prior to coating will vastly decrease the risk of outgassing during the coating process, in addition to mitigating the risk of delamination of the parylene. Outgassing can increase the length of time it takes to pump down the deposition chamber prior to coating, therefore increasing time-to-coat.
If water or other chemicals were in the product before it was coated, then as it heats up, bubbles will form under the coating, undermining its performance. Outgassing is the moisture or other chemical gases pushing out of a coated product underneath a coating, which can lead to bubbles forming. When the coating no longer adheres to the surface of the product, it can be susceptible to breaches or cracks forming from the inside due to internal pressures or from the outside due to mechanical abrasion or wear. Once the coating is breached, the product is open to contamination by corrosive chemicals.
Parylene Flammability
Flammability testing is an important component in product safety and compliance to a variety of industry standards. In flammability testing, parylene N will continue burning after a flame has been removed, so a product with N would have to rely on some other means for preventing a fire. The parylenes C, D, VT-4, and AF-4, contain halogens with C and D containing chlorine (Cl) and VT-4 and AF-4 containing fluorine (F), both of which function as flame retardants. This means that after a flame has been removed from these materials, they self-extinguish almost immediately.
Flammability is typically tested using UL 94 HB for horizontal burn and UL 94 V for vertical burn. Industries may call out their own specific test methods, but the UL 94 ones are very common, and are used for a product’s UL certification, as well as qualifying coating materials to IPC-CC-830 and MIL-I-46058 for high-reliability applications.
Cold Temperatures
Oxidation and degradation aren’t concerns at these low temperatures, but as you cool polymer materials down, including parylene, it becomes stiffer, less flexible, and more likely to incur physical damage. Parylene films can be flexed multiple times (up to 6 to 9) when cooled down to liquid nitrogen temperatures. Most other coatings will crack on first or second flex at these temperatures.
Wrapping It Up
Temperature ranges are one of the most important parameters when selecting the type of parylene that you’d like to use. If you have a challenging environment with higher and higher temperatures, you can expect the higher costs of the more exotic fluorinated parylenes, such as F and AF-4. By limiting the temperatures that your parylene-coated product will be exposed to, the more affordable options of N and C become available.
Which parylene to use should be considered early in the design phase, so that product engineering recommendations that may include design changes to the product are more likely to be considered and/or implemented, which in turn can improve the coating’s performance, the coated product’s reliability, and improve your company’s reputation. Whatever your needs, there’s a suitable parylene for products used in a variety of environments and conditions so long as you clearly convey those conditions early in the planning process.








