Why Parylene for Medical Device Coatings?

History of Approval.
Parylene has been used as a medical device coating for over four decades and is FDA-approved with a USP XXII, Class VI biocompatibility rating. 3rd party test certificates available on request.
Deposits as a Vapor.
Parylene film deposits one molecule at a time which enables parylene to coat intricate structures without pooling. Parylene is solvent-free, requires no curing and is deposited at room temperature.
Sterilize on any Substrate.
Parylene can be used with all common sterilization methods: e-beam, gamma, ethylene oxide (EtO) and autoclave. Ask about VSi’s parylene type F for high temperature requirements.
Next-Gen Material Properties.
Parylene’s unique material properties have allowed the medical device industry to advance innovative designs and find solutions for life saving technologies.
Medical Device Coating Applications
Barrier Layer
Advanced Barrier Protection for Medical Devices
Parylene provides a uniform, ultra-thin barrier against moisture, bodily fluids, electrical interference, and contaminants—enhancing device reliability with minimal added material or stress on components. From long-term implants to single-use electronics, parylene ensures durability and performance across a wide range of medical devices.
Often used in:
-
Medical PCBs and sensors
-
Robotic Laparoscopic Devices
-
Cochlear Implants
-
Pacemakers and Implantable
-
Cardioverter Defibrillator (ICD)
-
Orthopedic Hardware: Pins And Screws
Dielectric Layer
Precision Electrical Insulation for Medical Devices
Parylene provides a thin, uniform dielectric layer that insulates sensitive components, controlling current paths and preventing shorts or arcing. Its exceptional dielectric properties make it ideal for protecting electrically sensitive medical devices and ensuring reliable performance in critical applications.
Often used in:
-
Neurostimulation Devices
-
Ablation Devices
-
Electrodes
Dry Lubricity
Friction Reduction for Smoother Device Operation
Parylene N creates a thin, dry-film lubricant that lowers friction, enhancing device control and reducing trauma to blood vessels. Offering a low coefficient of friction similar to PTFE (Teflon) but without PFAS, parylene provides a safer, more environmentally friendly alternative without the risk of harmful chemical exposure or long-term environmental impact.
Often used in:
-
Endoscopic Cables
-
Silicone Guide Tubes
-
Catheters
-
Power and Communication Cables
-
Mandrels
-
Guide Wires
Tie Layer
Tie Layer for Stronger Coating Adhesion
Parylene acts as a biocompatible primer, improving adhesion between medical devices and drug coatings or lubricious layers. It supports both hydrophilic and hydrophobic coatings without reacting with carrier polymers or drugs. Parylene is non-thrombogenic and does not trigger an immune response, making it ideal for implantable and blood-contacting devices.
Often used in:
-
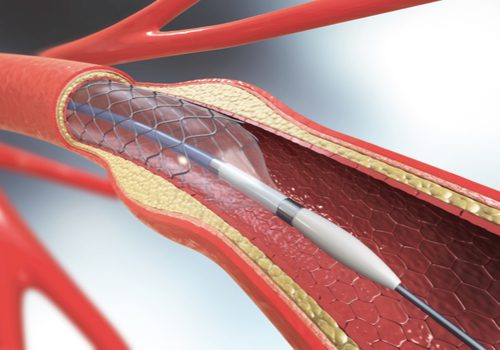
Catheters
-
Balloon catheters
-
Guidewires
-
Stents
-
Needles
Why VSi?
VSi Parylene is Focused on the Medical Device Industry.
VSi meets all the necessary vendor approval requirements for regulated medical device manufacturing. What sets us apart, however, is our dedication to solving the challenges that help our customers bring life-saving products to market.