Color and light are critical to our sense of sight and one of the most important ways that we interpret the world around us. Whether we’re viewing an ancient artifact to learn about our past, checking out an electronic billboard on our way to work, or discovering distant stars through a telescope, the parylenes are there helping us view and maintain our past, present, and future.
Like parylene’s other properties, the optical characteristics of parylene can be advantageous when planned appropriately. Parylene dimer starting materials begin the deposition process as a white to off-white powder and end as a clear, colorless coating, thus retaining desired color and clarity over time. That means you’ll always have a clear and accurate view.
General Optical Applications
Parylene coatings have been used for a wide variety of applications where the optical qualities have been critical to the coated object’s or product’s applications. Parylenes are used for environmental protection and maintaining the color accuracy and visual clarity of artwork and archival items of cultural and historical importance, such as those found in museums and galleries.
Parylenes protect both indoor and outdoor light-emitting diode (LED) displays used for advertising and entertainment. Outdoor applications can be challenging environments for the non-fluorinated parylenes, but these issues are not insurmountable.
Parylenes serve as protective coatings on optical lenses, electro-optical components, fiber optic components, optical sensors, and related products. Parylene has excellent transmission and conversely, low absorption, in the visible and near-infrared regions of the electromagnetic spectrum, which allows for very desirable performance in those regions.
Parylene UV, Vis, & NIR Absorption & Transmission Spectra
The ultraviolet (UV) – visible (Vis) – near-infrared (NIR) transmission spectra of parylenes N and C films are shown in Figure 1 below. The films were obtained from non-purified monomers and show a good transmittance in the visible domain from 390 nm (violet) to 750 nm (red), so that the films appear colorless.
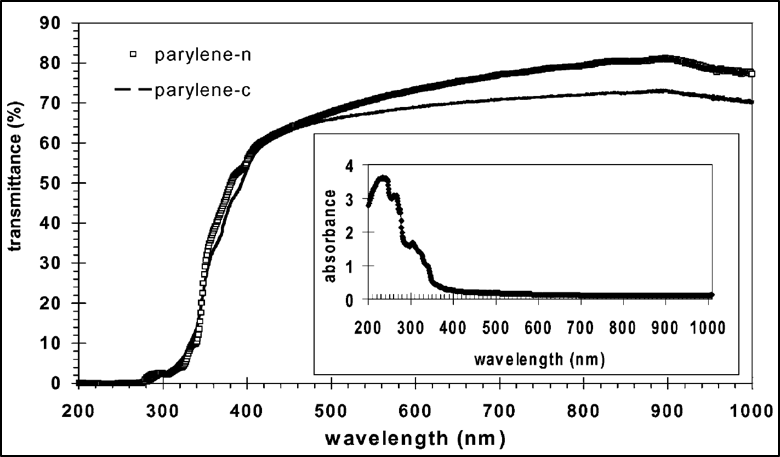
Figure 1. UV-Vis-NIR transmission and absorbance spectra of parylenes N and C self-supported films.
Parylene UV Resistance, Transmission & Absorption – Interactions with Ultraviolet Light
Extended exposure to UV will degrade parylenes N, C, D, and much later, parylene F. With these coatings, direct outside usage will be of limited value, unless measures are taken to limit direct sunlight exposure. If you limit the amount of oxygen near the coating, then UV exposure will not be as detrimental since the presence of both is required to cause significant issues.
Note though, that parylene F is better than N and C, but the starting material can cost three to five times more than C. More significantly though, parylene AF-4 is much better than parylene F at resisting UV damage, but the starting material can cost up to twenty times more than C and has very limited availability and suppliers. A graph comparing the UV exposure performance of parylene N, C, and F is shown in Figure 2 below.
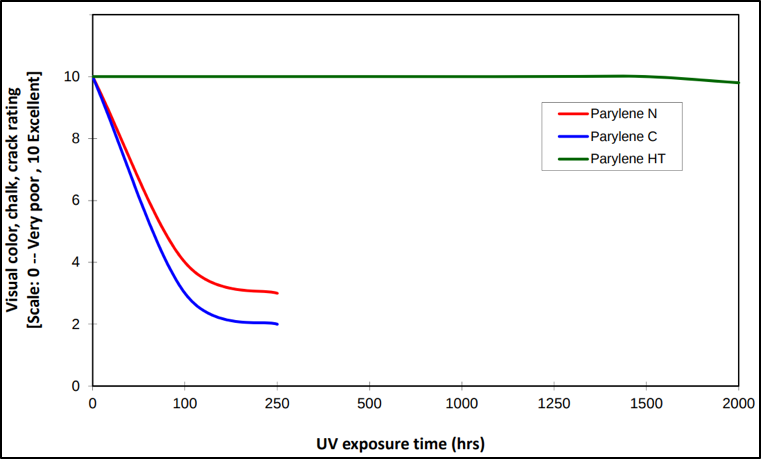
Figure 2. UV stability graph comparing physical appearance changes versus exposure time for C, N, and AF-4.
The degradation pathway for the non-fluorinated (non-AF-4) parylenes follows the reaction scheme shown in Figure 3. Based on information from Bera and others, the degradation products act as UV absorbers, which limit the amount of UV that reaches further into the thin film. Along with the slightly lower oxygen permeability of parylene C versus N, the retention of mechanical properties as the film yellows is better for parylene C versus N. Eventually, as the polymer oxidizes, main-chain scission occurs, creating breaks in the polymer at the molecular level, and leading to cracks at the bulk level.
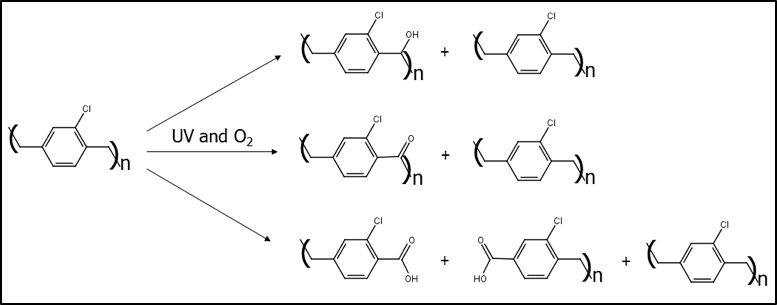
Figure 3. Degradation reaction scheme for Parylene C’s oxidation in the presence of UV and oxygen.
Protective coatings, including parylenes, work best when they’re free of pinholes, bubbles, and most importantly cracks. Exposure to ultraviolet (UV) light, especially from the sun, and oxygen causes microscopic breaks in most parylene films until it is severe enough to cause cracks visible to the naked eye.
In cases of UV light or high temperature and oxygen, the more commonly used parylenes become oxidized. Carbon to oxygen bonds are added to the methylene (CH2) units in the polymer backbone: first as an alcohol (-OH), then a carbonyl (C=O), and then a carboxylic acid (-COOH) which causes a break in the polymer chain. When enough of the polymer chains have broken in such a manner, then a visible crack will form.
Once a crack has formed, the product will be exposed to possible contamination and corrosion. Since this oxidation and degradation mechanism involves CH2 units, then the parylenes N, C, D, and F are susceptible. Only parylene AF-4 has CF2 units bridging between the rings in the polymer chain and that’s why it can survive much higher temperatures and much longer UV exposure than the other parylenes.
Transparent in the Visible Region
Being colorless is important for protecting light-emitting devices, since the parylene coatings must not affect the color of the emitted light nor the device efficiency. Thus, parylenes are useful for cameras and optical devices exposed to harsh environments such as traffic cameras and speed sensors.
Transparent in the Near-Infrared Region (NIR)
Parylene works well in products that use touch screens based on infrared (IR) emitters and detectors. Examples of how these screens work are shown in the diagrams featured in Figures 4 and 5 below.
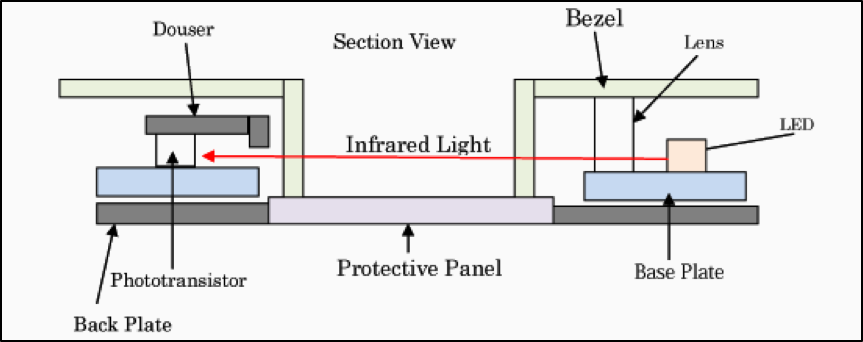
Figure 4. Cross-sectional diagram of IR touch screen technology.
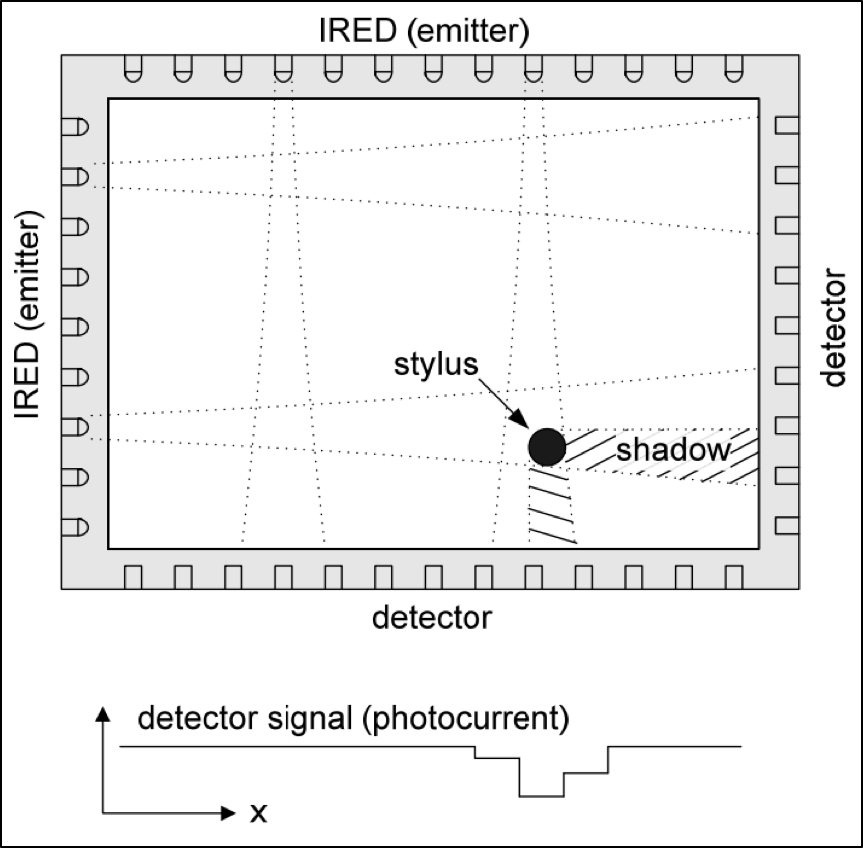
Figure 5. Overview diagram and electrical signal concept of IR touch screen technology.
Parylene Refractive Index
The refractive index of a material is a ratio of the speed of light in a material compared to the speed of light in a vacuum. When light passes through a material, atoms are constantly absorbing and re-emitting particles of light which slows it down. The refractive indices for the common parylenes are listed in Table 1 below. Parylene N’s refractive index of 1.66 means that the speed of light in N is 1.66 times slower than light in a vacuum.
Table 1. Refractive indices for the common parylenes.
| Properties | Parylene N | Parylene C | Parylene D | Parylene F | Parylene AF-4 |
| Index of Refraction (nD23) | 1.66 | 1.64 | 1.67 | 1.57 | 1.56 |
Refractive indices are important in many optical applications since they’re used to calculate the focusing power of lenses, the dispersive power of prisms, and to describe how much of a change in direction the light will have as it passes from one medium into another.
Wrapping It Up
Other than direct UV exposure for the non-fluorinated types, the parylenes provide beneficial optical properties for many types of applications ranging from protecting museum artifacts to opto-electronic components that help enhance the reliability of aerospace, scientific, and telecommunication devices. It’s clear to see, when protecting products that require color and clarity to be maintained, parylene is an excellent choice.
FAQs
How does parylene protect artifacts from UV light?
Parylene coatings form a protective barrier over the object, blocking out UV light and thus protecting the object from degradation or damage. Parylene F and parylene AF-4 are the most resistant to UV damage, however the starting material for AF-4 can cost up to twenty times more than parylene C.
What is the optical transmittance of parylene?
Parylene coatings have a good transmittance in the visible domain from 390 nm (violet) to 750 nm (red), so that the films appear colorless. This allows for very desirable performance in the ultraviolet (UV) – visible (Vis) – near-infrared (NIR) regions of the electromagnetic spectrum.
What is the refractive index of parylene?
The refractive index of a material is a ratio of the speed of light in a material compared to the speed of light in a vacuum. The refractive indices for the common parylenes are listed in Table 1 below. Parylene N’s refractive index of 1.66 means that the speed of light in N is 1.66 times slower than light in a vacuum.
How is parylene used in light-emitting devices?
Parylenes are used for environmental protection and maintaining the color accuracy and visual clarity of artwork and archival items of cultural and historical importance, such as those found in museums and galleries. Parylene coatings also protect LED displays used for advertising and entertainment, and are used as protective coatings on optical lenses, electro-optical components, fiber optic components, optical sensors, and related products.








